
Dr. Alessia Ruggieri
Alessia.Ruggieri
(at)med.uni-heidelberg.de
Phone: +49 6221 56–7761
Fax: +49 6221 56–4570
Translational Control by RNA Viruses
Projects
Throughout the infection, viruses elicit multiple host cell responses including innate immune and stress responses. Viral double-stranded (ds) RNA replication intermediates trigger the activation of the stress sentinel Protein kinase R (PKR) which mediates the phosphorylation of eIF2α, a critical factor for translation initiation. As an almost immediate result, polysomes disassemble, protein synthesis is suppressed. Stalled mRNAs condensate with RNA-binding proteins and form membrane-less stress granules (SGs) which formation is dynamic, reversible and driven by cytosolic phase separation. To establish productive infections, viruses have evolved mechanisms to overcome translational attenuation that results from stress response induction. My laboratory is trying to understand how RNA viruses control the host translation machinery and the cellular stress response to ensure their progeny production. We explore the strategies evolved by different members of the Flaviviridae family such as hepatitis C virus (HCV), dengue virus (DENV), Zika virus (ZIKV) and West Nile virus (WNV) to antagonize or inversely to utilize the host stress response pathway.
Reasearch Interests:
Host Stress Response to RNA Viruses
Stress Granules
Translational Repression by Viruses
Innate Sensing of Retroviruses
1 | Dynamic Stress Response to Hepatitis C Virus Infection
Using long-term live-cell imaging microscopy, we showed that HCV infection in combination with type I interferon (IFN) treatment induces a dynamic and oscillating host cell stress response that can be visualized by cycles of assembly and disassembly of SGs (Ruggieri et al., 2012). We recently addressed the complex regulation of this dynamic process with help of quantitative mathematical modelling and are currently investigating the biological function of SGs in HCV chronic infection. Our previous work was performed using two-dimensional cultures of hepatoma-derived Huh7 cells that are proliferative and have immune-competence and lost their main hepatic functions. We are currently setting up three-dimensional hepatocyte-like cultures which is compatible with long-term live cell imaging and light-sheet microscopy to investigate HCV-induced stress response under more physiological conditions.
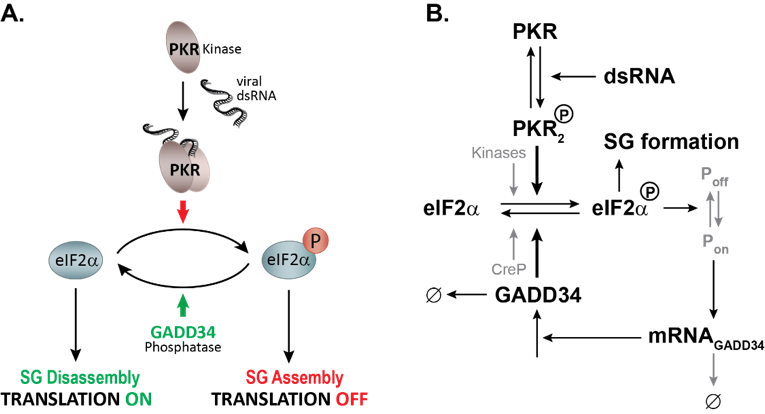
Figure 1. Mathematical modeling of HCV-induced SG oscillations. A. Description of the stress signaling pathway activated by HCV infection. PKR is activated by binding to viral dsRNA, dimerizes and autophosphorylates. Active PKR phosphorylates its direct substrate eIF2α leading to a bulk translation initiation shutoff, polysome disassembly and assembly of SGs. To promote survival, cells GADD34 is transcriptionally and translationally upregulated and in complex with the Protein Phosphatase 1 dephosphorylates eIF2α. Translation initiation is reactivated and SGs disassemble. As long as viral dsRNA is present in the cell, these cycles of active and stalled translation will occur. B. Mathematical model of SG oscillations.
2 | Regulation of GADD34, the Stress-induced Regulatory Subunit of Phosphatase PP1
HCV-induced oscillating SG formation is regulated at the level of the eukaryotic initiation factor 2 alpha (eIF2α) by the antagonistic action of two main switches, PKR and GADD34, the stress-induced regulatory subunit of Protein Phosphatase 1. The mathematical model of oscillating SGs identified GADD34 as an important and dynamic node of regulation, both at the protein and at mRNA levels. We experimentally validated that under normal conditions GADD34 is a short-lived protein whose mRNA exhibits a rapid turnover and are currently investigating the signaling pathways responsible for GADD34 regulation upon stress induction.
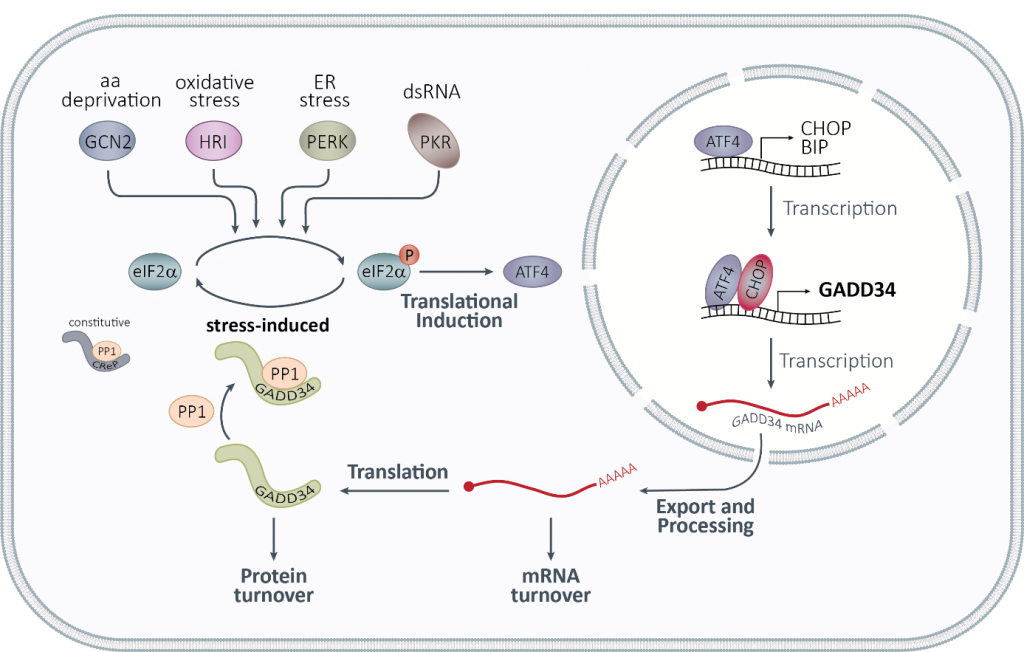
Figure 2. The integrated stress response.
3 | Translational Control by Flaviviruses
Flaviviruses such as DENV, ZIKV and WNV have a (+) single-stranded RNA genome with a type I cap at their 5’ end and a non-polyadenylated 3′ untranslated region. Viral genomes therefore compete with host mRNAs for their translation. We have recently shown that flavivirus infection induces a severe repression of the host cell translation in human cells, which is uncoupled from the cellular stress response (Roth et al., 2017). Importantly, translation of viral genomes is maintained while host translation is repressed. This work suggested an unconventional and virus-specialized translation initiation mechanism that we are currently investigating.
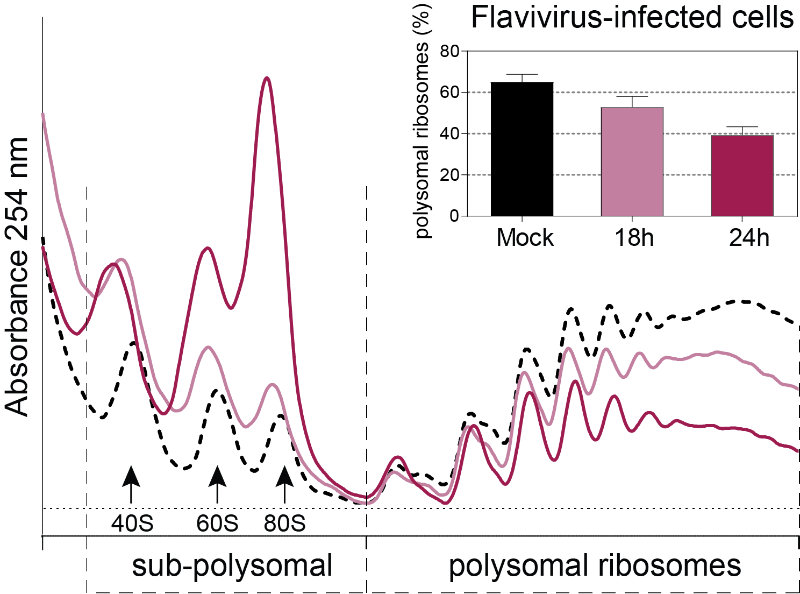
Figure 3. Polysome profiles of Flavivirus-infected Huh7 cells. This technique allows the identification of translational changes in host cells submitted to different environmental stresses including virus infection by separating heavier ribosome-associated mRNAs (actively translating mRNAs, polysomal ribosomes) from the light sub-polysomal mRNAs (Poorly or not translated mRNAs, sub-polysomal ribosomes). Flaviviruses reduce the bulk of active translating mRNAs in the course of the infection.
4 | Role of TREX1 for the Innate Sensing of Retroviruses
As a negative regulator of innate immunity, Three Prime Repair Exonuclease 1 (TREX1) acts in a dual function to protect against autoimmune phenotypes, such as Aicardi-Goutières syndrome (AGS), familial chilblain lupus (FCL), systemic lupus erythematosus (SLE) and retinal vasculopathy with cerebral leukodystrophy (RVCL). TREX1 is the most abundant 3’-5’ DNA exonuclease in mammalian cells, which main function as part of the SET complex in the nucleusis to process aberrant single- and double-stranded DNA replication intermediates that accumulate during DNA replication. On the other hand, TREX1 also localizes at the endoplasmic reticulum where it stabilizes the catalytic activity of the oligosaccharyltransferase (OST) complex. Recently, TREX1 also shown to metabolize exogenous retroviral reverse transcription (RT) products in the cytoplasm of mammalian cells and therefore suggested to play a potential role in the elimination of endogenous retrovirus RT products. Unlike in mice, several splice variants of TREX1 are detected in human cells. We are currently investigating TREX1 isoform expression and regulation in human peripheral blood-derived cells, as well as their localization and exonuclease function, particularly in the context of retroviral infection.
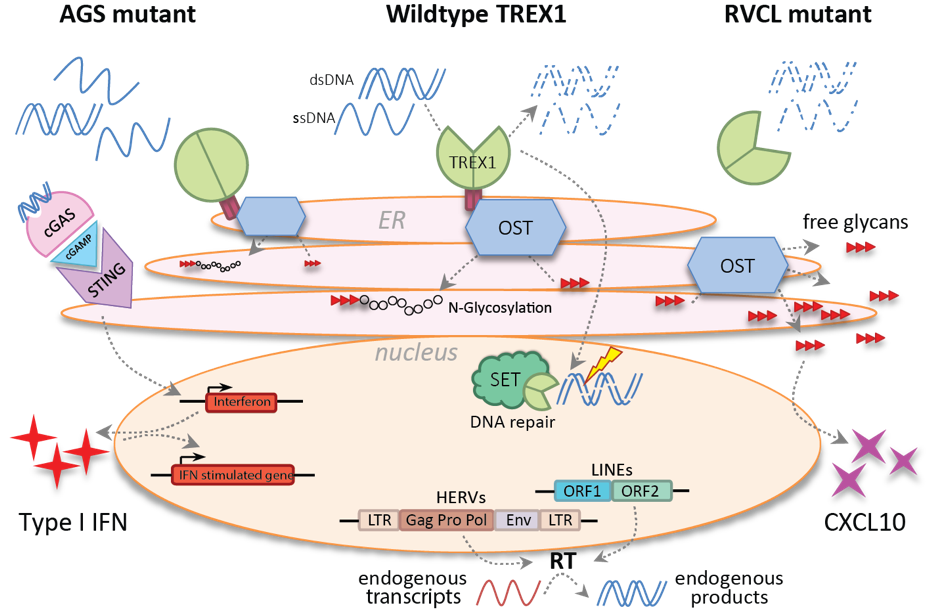
Figure 4. The multiple roles of TREX1 in mammalian cells.
- Ruggieri Lab Molecular Virology:
https://www.klinikum.uni-heidelberg.de/zentrum-fuer-infektiologie/molecular-virology/about-us/research-teams/ag-ruggieri - SFB1129: https://www.sfb1129.de/projects/project-13/project-13–2nd-funding-period/
- TRR186: https://trr186.uni-heidelberg.de/en/node/27
- SPP1923: https://spp1923.de/projects/ruggieri-fackler/
Complete Publication List (PubMed)
ORCID-iD: 0000–0001-9981–3308
Ruggieri A, Helm M, Chatel-Chaix L. An epigenetic “extreme makeover”: the methylation of flaviviral RNA (and beyond). 2020. RNA Biol 18:1–13.
Eiermann N, Haneke K, Sun Z, Stoecklin G, Ruggieri A. Dance with the devil: Stress granules and signaling in antiviral responses. 2020. Viruses 12(9), 984.
Haneke K, Schott J, Lindner D, Hollensen AK, Damgaard CK, Mongis C, Knop M, Palm W, Ruggieri A, Stoecklin G. CDK1 couples proliferation with protein synthesis. 2020. J Cell Biol. 219(3): e201906147.
Brocard M, Iadevaia V, Klein P, Hall B, Lewis G, Lu J, Burke J, Willcocks M, Parker R, Goodfellow IG, Ruggieri A, Locker N. Norovirus infection results in eIF2α-independent host translation shut-off and remodels the G3BP1 interactome evading stress granule formation. 2020. PLoS Pathog. 16(1):e1008250.
Ruggieri, A. and Stoecklin, G. (2019). A Signal to Condense. Nat Chem Biol. 15(1):5–6.
Schult, P., Roth H., Adams, R.L., Mas, C., Imbert, L., Orlik, C., Ruggieri, A., Pyle, A.M., Lohmann, V. (2018). MicroRNA-122 amplifies hepatitis C virus translation by shaping the structure of the internal ribosomal entry site. Nat Commun. 9(1):2613.
Roth, H., Magg, V., Uch, F., Mutz, P., Klein, P., Haneke, K., Lohmann, V., Bartenschlager, R., Fackler, O.T., Locker, N., Stoecklin, G., Ruggieri, A. (2017). Flavivirus infection uncouples translation suppression from cellular stress responses. mBio 8(1):e02150-16.
Brocard, M., Ruggieri, A., Locker, N. (2017). m6A RNA methylation, a new hallmark in virus-host interactions. J Gen Virol. 98(9):2207–2214.
Cortese, M., Goellner, S., Acosta, E.G., Neufeldt, C.J., Oleksiuk, O., Lampe, M., Haselmann, U., Funaya, C., Schieber, N., Ronchi, P., Schorb, M., Pruunsild, P., Schwab, Y., Chatel-Chaix, L., Ruggieri, A., Bartenschlager, R. (2017). Ultrastructural characterization of Zika virus replication factories. Cell Reports 18(9): 2113–2123.
Trautz, B., Wiedemann, H., Lüchtenborg, C., Pierini, V., Kranich, J., Glass, B., Kräusslich, H.-G., Brocker, T., Pizzato, M., Ruggieri, A., Brügger, B., Fackler, O.T. (2017). The host-cell restriction factor SERINC5 restricts HIV‑1 infectivity without altering the lipid composition and organization of viral particles. J Biol Chem 292(33):13702–13713.
Trotart, M., Tsopoulidis, N., Tibroni, N., Willemsen, J., Binder, M., Ruggieri, A., Fackler, O.T. (2015). Sensing of HIV‑1 infection in Tzm-bl cells with reconstituted expression of STING. J Virol. 90(4):2064–76.
Schmid, B., Rinas, M., Ruggieri, A., Reuter, A., Fischl, W., Harder, N., Bergeest, J.-P., Flossdorf, M., Rohr, K., Höfer, T., Bartenschlager, R. (2015). Live-cell analysis and modeling identify determinants of attenuation of Dengue virus 2‑O-methyl mutant. PLoS Pathog. 11(12):e1005345.
Hiet, M.-S., Bauhofer, O., Zayas, M., Roth, H., Tanaka, Y., Schirmacher, P., Willemsen, J., Grünvogel, O., Bender, S., Binder, M., Lohmann, V., Lotteau, V., Ruggieri, A.†, Bartenschlager, R†. (2015). Control of temporal activation of hepatitis C virus-induced interferon response by domain 2 of nonstructural protein 5A. J Hepatol. 63(4):829–37.
Ruggieri, A., Dazert, E., Metz, P., Hofmann, S., Bergeest, J.-P., Mazur, J., Bankhead, P., Hiet, M.-S., Kallis, S., Alvisi, G., Samuel, C.E., Lohmann, V., Kaderali, L., Rohr, K., Frese, M., Stoecklin, G., Bartenschlager, R. (2012). Dynamic oscillation of translation and stress granule formation mark the cellular response to virus infection. Cell Host Microbe 12(1): 71–85.
